-
PDF
- Split View
-
Views
-
Cite
Cite
Alec Saitman, Hyung-Doo Park, Robert L. Fitzgerald, False-Positive Interferences of Common Urine Drug Screen Immunoassays: A Review, Journal of Analytical Toxicology, Volume 38, Issue 7, September 2014, Pages 387–396, https://doi.org/10.1093/jat/bku075
Close - Share Icon Share
Abstract
Urine drug screen (UDS) immunoassays are a quick and inexpensive method for determining the presence of drugs of abuse. Many cross-reactivities exist with other analytes, potentially causing a false-positive result in an initial drug screen. Knowledge of these potential interferents is important in determining a course of action for patient care. We present an inclusive review of analytes causing false-positive interferences with drugs-of-abuse UDS immunoassays, which covers the literature from the year 2000 to present. English language articles were searched via the SciFinder platform with the strings ‘false positive [drug] urine’ yielding 173 articles. These articles were then carefully analyzed and condensed to 62 that included data on causes of false-positive results. The discussion is separated into six sections by drug class with a corresponding table of cross-reacting compounds for quick reference. False-positive results were described for amphetamines, opiates, benzodiazepines, cannabinoids, tricyclic antidepressants, phencyclidine, lysergic acid diethylamide and barbiturates. These false-positive results support the generally accepted practice that immunoassay positive results are considered presumptive until confirmed by a second independent chemical technique.
Introduction
Immunoassays dominate urine drug screens (UDSs) because they are simple to use, easy to automate and provide rapid results (1). Unfortunately, they are subject to cross-reactivity with structurally related and unrelated compounds potentially yielding false-positive results. Further complicating the issue are the many available platforms with differing cross-reactivities. Immunoassays for selected drug classes, e.g., opiates and benzodiazepines, are also subject to clinically important false negatives (2). False-negative results can be caused by a variety of factors including the cross-reactivity of the antibody used by the assay, the cutoff concentration for a positive result and length of time between drug ingestion and specimen acquisition. False negatives are not covered in this review but present opportunities for significant patient mismanagement if not understood. The best practice following a positive UDS involves confirmation with the mass spectrometry (MS) technique such as gas chromatography–mass spectrometry (GC-MS) or liquid chromatography–tandem mass spectrometry. Regrettably, MS testing is limited or nonexistent in many hospital laboratories. When confirmatory testing is performed, results are generally unavailable for several days. Owing to the delay in receiving confirmation results, decisions about patient care are frequently made on ‘presumptive positive’ drug screening results.
Because many providers have limited knowledge of immunoassay cross-reactivity data, patients with false-positive results may lose eligibility in rehabilitation programs, be inappropriately terminated from employment or suffer from medical staff bias because of lack of trust (3). Although this topic has been reviewed previously (2–4), our aim is to provide a concise, comprehensive and up-to-date account of substances potentially interfering with UDS immunoassays. This review is meant to serve as a guide for practitioners to assess potential false-positive UDS results while waiting for confirmatory test results to become available.
This review focuses on cases where the cause of a false positive is identified. In practice, there are many cases where immunoassay positive results are not confirmed when analyzed with a more sensitive and specific technique. While it is useful to understand some of the common causes of false-positive immunoassay results, in the majority of cases the cause of false-positive screening results is unknown. Owing to the number of false positives, all immunoassay results are considered ‘presumptive’ until confirmed by an independent chemical technique.
Materials and methods
An inclusive literature review was conducted for five predefined drugs of abuse classes and a miscellaneous drug class that included several different drugs. The predefined drug classes include amphetamines, tricyclic antidepressants (TCAs), benzodiazepines, cannabinoids and opiates (natural and synthetic). The drug classes were then investigated using the SciFinder platform that includes MEDLINE and CAPLUS databases. The strings ‘false positive [drug] urine’ for English language articles were searched. In addition, a panel of over 60 common over the counter medications, prescription drugs and their metabolites, and illicit drugs were included using the same search syntax. To maintain current relevance, only articles published after the year 2000 were included in the search. This search yielded 173 articles. These articles were then analyzed to determine if they included data on false-positive urine immunoassay results. A total of 62 articles met these criteria and were included in this review. Any articles prior to 2000 included in this review are used to clarify current cross-reactivity issues.
Publications describing interferences with UDS present differing degrees of evidence to support their claims. The weakest evidence is noted by simply stating that the patient(s) had exposure to the proposed interfering drug, or comparing two-dimensional drug structures for similarity or dissimilarity between the proposed interferent and the assay target drug/drug class. Stronger evidence is obtained by adding a pure standard of the suspected interferent drug (and/or potential metabolites) at several concentrations to drug free urine (DFU) and testing the immunoassay(s) in question. Weaker variants of this protocol include adding crushed tablets or capsules instead of pure drug standard and using water or organic solvent/water mixtures instead of human DFU as a matrix. Useful supportive evidence may include quantitation of potentially interfering substances in patient specimens and chart reviews, but these methods are not a substitute for directly testing the proposed interferent in the appropriate matrix. Some studies dose subjects with the drug in question and evaluate immunoassays before and after dosing. In these cases, positive results are analyzed by confirmatory testing methods to demonstrate the absence of the target compound. The articles presented here for drugs causing false-positive immunoassay results include all of the above methods.
Amphetamines
False-Positive Results for Amphetamine Immunoassays
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Bupropion | Siemens Dimension® | SYVA EMIT II Plus® | Occurred | Retrospective chart review | (6) |
| Chlorpromazine | Roche Hitachi 911® (1.0 μg/mL cutoff) | SYVA EMIT-MAM® | Occurred | Retrospective chart review | (1) |
| DMAA | Roche Modular P® (300 ng/mL cutoff) | Roche kinetic interaction of microparticles in a solution (KIMS®) | 7.5 μg/mL | Spiking of standards in DFU | (7) |
| SYVA EMIT II Plus® | 3.1 μg/mL | Spiking of standards in DFU | (7) | ||
| Roche kinetic interaction of microparticles in a solution (KIMS)® and SYVA EMIT II® | 6.9 μg/mL | Therapeutic dosing | (7) | ||
| Labetalol | Not mentioned | Not mentioned | Occurred | Case report | (8) |
| Metformin | – | Biosite Triage® | Occurred | Case report | (9) |
| Ofloxacin | TdxFlx® (300 ng/mL cutoff) | AM/MA II® | Occurred | Therapeutic dosing | (10) |
| Promethazine | Roche Hitachi 911® (1.0 μg/mL cutoff) | SYVA EMIT-MAM® | Occurred | Retrospective chart review | (1) |
| Trazodone (m-CPP) | Olympus UA 400® | SYVA Ecstasy EMIT II® | 3 μg/mL (m-CPP) | Spiking of standards in DFU | (11) |
| Roche Cobas c501®, cutoff 300 μg/L | Amphetamines II® | 6.7 μg/mL (m-CPP) | Native matrix | (12) |
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Bupropion | Siemens Dimension® | SYVA EMIT II Plus® | Occurred | Retrospective chart review | (6) |
| Chlorpromazine | Roche Hitachi 911® (1.0 μg/mL cutoff) | SYVA EMIT-MAM® | Occurred | Retrospective chart review | (1) |
| DMAA | Roche Modular P® (300 ng/mL cutoff) | Roche kinetic interaction of microparticles in a solution (KIMS®) | 7.5 μg/mL | Spiking of standards in DFU | (7) |
| SYVA EMIT II Plus® | 3.1 μg/mL | Spiking of standards in DFU | (7) | ||
| Roche kinetic interaction of microparticles in a solution (KIMS)® and SYVA EMIT II® | 6.9 μg/mL | Therapeutic dosing | (7) | ||
| Labetalol | Not mentioned | Not mentioned | Occurred | Case report | (8) |
| Metformin | – | Biosite Triage® | Occurred | Case report | (9) |
| Ofloxacin | TdxFlx® (300 ng/mL cutoff) | AM/MA II® | Occurred | Therapeutic dosing | (10) |
| Promethazine | Roche Hitachi 911® (1.0 μg/mL cutoff) | SYVA EMIT-MAM® | Occurred | Retrospective chart review | (1) |
| Trazodone (m-CPP) | Olympus UA 400® | SYVA Ecstasy EMIT II® | 3 μg/mL (m-CPP) | Spiking of standards in DFU | (11) |
| Roche Cobas c501®, cutoff 300 μg/L | Amphetamines II® | 6.7 μg/mL (m-CPP) | Native matrix | (12) |
False-Positive Results for Amphetamine Immunoassays
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Bupropion | Siemens Dimension® | SYVA EMIT II Plus® | Occurred | Retrospective chart review | (6) |
| Chlorpromazine | Roche Hitachi 911® (1.0 μg/mL cutoff) | SYVA EMIT-MAM® | Occurred | Retrospective chart review | (1) |
| DMAA | Roche Modular P® (300 ng/mL cutoff) | Roche kinetic interaction of microparticles in a solution (KIMS®) | 7.5 μg/mL | Spiking of standards in DFU | (7) |
| SYVA EMIT II Plus® | 3.1 μg/mL | Spiking of standards in DFU | (7) | ||
| Roche kinetic interaction of microparticles in a solution (KIMS)® and SYVA EMIT II® | 6.9 μg/mL | Therapeutic dosing | (7) | ||
| Labetalol | Not mentioned | Not mentioned | Occurred | Case report | (8) |
| Metformin | – | Biosite Triage® | Occurred | Case report | (9) |
| Ofloxacin | TdxFlx® (300 ng/mL cutoff) | AM/MA II® | Occurred | Therapeutic dosing | (10) |
| Promethazine | Roche Hitachi 911® (1.0 μg/mL cutoff) | SYVA EMIT-MAM® | Occurred | Retrospective chart review | (1) |
| Trazodone (m-CPP) | Olympus UA 400® | SYVA Ecstasy EMIT II® | 3 μg/mL (m-CPP) | Spiking of standards in DFU | (11) |
| Roche Cobas c501®, cutoff 300 μg/L | Amphetamines II® | 6.7 μg/mL (m-CPP) | Native matrix | (12) |
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Bupropion | Siemens Dimension® | SYVA EMIT II Plus® | Occurred | Retrospective chart review | (6) |
| Chlorpromazine | Roche Hitachi 911® (1.0 μg/mL cutoff) | SYVA EMIT-MAM® | Occurred | Retrospective chart review | (1) |
| DMAA | Roche Modular P® (300 ng/mL cutoff) | Roche kinetic interaction of microparticles in a solution (KIMS®) | 7.5 μg/mL | Spiking of standards in DFU | (7) |
| SYVA EMIT II Plus® | 3.1 μg/mL | Spiking of standards in DFU | (7) | ||
| Roche kinetic interaction of microparticles in a solution (KIMS)® and SYVA EMIT II® | 6.9 μg/mL | Therapeutic dosing | (7) | ||
| Labetalol | Not mentioned | Not mentioned | Occurred | Case report | (8) |
| Metformin | – | Biosite Triage® | Occurred | Case report | (9) |
| Ofloxacin | TdxFlx® (300 ng/mL cutoff) | AM/MA II® | Occurred | Therapeutic dosing | (10) |
| Promethazine | Roche Hitachi 911® (1.0 μg/mL cutoff) | SYVA EMIT-MAM® | Occurred | Retrospective chart review | (1) |
| Trazodone (m-CPP) | Olympus UA 400® | SYVA Ecstasy EMIT II® | 3 μg/mL (m-CPP) | Spiking of standards in DFU | (11) |
| Roche Cobas c501®, cutoff 300 μg/L | Amphetamines II® | 6.7 μg/mL (m-CPP) | Native matrix | (12) |
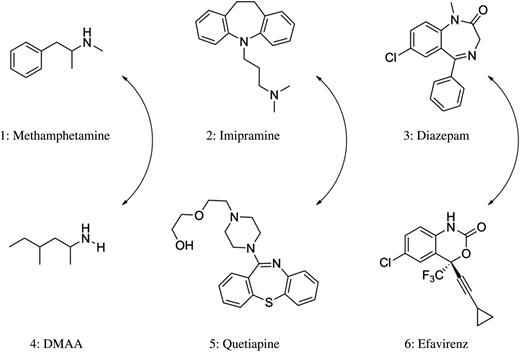
Structures of methamphetamine, imipramine and diazepam (top) along with cross-reacting compounds (bottom).
Dimethylamylamine (DMAA), a widely used energy supplement, causes false-positive amphetamine screens in both the Roche KIMS® and SYVA EMIT II® kits on a Roche/Hitachi Modular P platform (7). With spiking experiments, Vorce et al. demonstrated that concentrations of 3.1 μg/mL of DMAA (4) analyzed on the EMIT II® assay caused a false-positive result for amphetamines (Figure 1). The KIMS® assay was more specific, requiring concentrations of 7.5 μg/mL and higher for a false-positive result. A separate examination was completed using urine samples from patients taking DMAA. When samples from patients taking DMAA were tested, concentrations at or >6.9 μg/mL returned false-positive results for both kits (Table I) (7).
Yee and Wu (8) described three reports of women prescribed labetalol testing positive for amphetamines. GC–MS analysis of these specimens failed to detect amphetamine or methamphetamine. The authors cite reports of a metabolite of labetalol, 3-amino-1-phenylbutane being known to cross-react with multiple amphetamine immunoassays (8).
Bupropion has also been shown to cross-react with amphetamine immunoassays. Casey et al. (6) completed a retrospective chart review of positive UDS amphetamine results which failed to confirm by GC–MS. Forty-one percent of these samples (53 patients) were from patients taking bupropion at the time of the UDS. After statistically ruling out poly-substance abuse as a cause, the investigators concluded that a large portion of these false-positive outcomes stemmed directly from the patients' bupropion intake (6). An important limitation of this study was the lack of spiking experiments demonstrating bupropion (or associated metabolites) truly causing the false-positive result (Table I).
An investigation by Melanson et al. (1) reveals that the structurally similar medications promethazine and chlorpromazine, both used to treat psychiatric conditions, can account for false-positive amphetamine UDSs . All patients with promethazine or chlorpromazine detected in their serum by liquid chromatography–photo-diode array (LC–PDA) detection, who also had an SYVA EMIT-MAM® (monoclonal amphetamine/methamphetamine) urine toxicology screen analyzed on the Roche Hitachi 911® platform, regardless of the results, were included in the study (Table I). A presumptive positive amphetamine result was considered if the sample was found to be above the cutoff concentration of the lowest calibrator for the assay, which is 1.0 μg/mL (1). Three urine specimens from patients taking promethazine were positive on the EMIT-MAM® assay, and all three were negative for amphetamines by LC–PDA. The authors also describe six false-positive amphetamine results in patients taking chlorpromazine that were negative for amphetamines via LC–PDA (Table I). The authors then analyzed all nine of these promethazine/chlorpromazine urine specimens on three other amphetamine UDS kits including SYVA EMIT-A®, Biosite® Triage and Agilent™ TesTcard 9. The results of the three promethazine and six chlorpromazine patient samples were all negative via the SYVA EMIT-A® and Biosite® Triage assays. However, the TesTcard 9 was subject to two false-positive results, one occurring from a patient taking promethazine and the other from a patient taking chlorpromazine (1). The authors conclude that both promethazine and chlorpromazine can cause false-positive amphetamines UDS and recommend confirmation of any presumptive positive results with a secondary method (1).
The commonly prescribed antidepressant, trazodone, has been reported to cause false positives in 3,4-methylenedioxymethamphetamine (MDMA) UDSs. A study conducted by Logan et al. (11) describes trazodone specifically cross-reacting with the EMIT II Plus® Ecstasy polyclonal assay, but not with the EMIT II Plus® Amphetamine monoclonal kit. The study consisted of spiking trazodone standards into DFU, then running the samples with both assay kits on the Olympus U400® platform (Table I). The authors concluded that trazodone can cause false-positive MDMA results at 3.0 μg/mL. The authors hypothesize that this false-positive result is most likely due to a metabolite of trazodone, meta-chlorophenylpiperazine (m-CPP), owing to its structural similarity with MDMA (11).
Baron et al. performed dilution experiments of m-CPP and trazodone standards in water and showed that false positives also occurred on the Amphetamine II assay on the Roche Cobas c501®. They demonstrated that a positive result for amphetamines will occur at concentrations of m-CPP at or >6.7 μg/mL when using a 1.0 μg/mL d-methamphetamine as the cutoff calibrator. These authors also showed that the concentration of trazodone would have to be >200 μg/mL to yield the positive result (12). The authors then inspected six patients with confirmed positive serum trazodone results by LC–MS and compared the results with the findings of the UDSs run on the aforementioned assay. Three patients with a trazodone concentration of >1.0 μg/mL and a serum m-CPP concentration of >0.85 μg/mL returned a positive result for amphetamines with the Amphetamines II® method (12). These presumptive positives were then confirmed negative for amphetamines via GC–MS (Table I).
Nomier and AL-Huseini (10) demonstrated that the Amphetamine/Methamphetamine II® kit run on the TdxFlx® platform can generate false-positive amphetamine results in the presence of ofloxacin . Urine samples were collected before and after administration of two 200 mg ofloxacin doses in six healthy male volunteers at an interval of 12 h. The urine samples were then analyzed using the Amphetamine/Methamphetamine II® assay on the TdxFlx. No samples prior to ofloxacin administration yielded a positive result for amphetamines. After administration however, all six samples returned positive results for amphetamines, the lowest of which was almost four times the concentration of the calibrator cutoff of 300 ng/mL (Table I) (10).
Tricyclic antidepressants
TCAs are a class of drugs mainly used to treat anxiety, eating disorders and depression (13). As the name suggests this class of compounds is based on a three-ringed organic framework, which is further modified to obtain desired pharmacological effects. The TCA assays have historically had a high rate of false positives. Drugs and/or metabolites described at or prior to the year 2000 as causing false-positive TCA results include carbamazepine (14) and cyclobenzaprine (15). Our literature search of recent interferences revealed that quetiapine (5), usually prescribed as an atypical antipsychotic (16), was the only recent drug found to yield false-positive TCA results (Figure 1).
In one report, the investigators created dilutions of quetiapine tablets dissolved in water and demonstrated that false positives for TCAs were occurring at 7.0 μg/mL using the Microgenics® Tricyclics Serum Tox EIA immunoassay on the Roche™ Hitachi 911® platform (16). A similar study demonstrated that quetiapine caused false-positive TCA results with the Microgenics® immunoassay on the Beckman™ LX20® platform and with the Syva® RapidTest d.a.u.® point-of-care (POC) kit (Table II). False positives occurred at 10.0 and 100.0 μg/mL, respectively (17). These investigators also analyzed urine from a patient prescribed a therapeutic dose of quetiapine 4 h prior to sample collection. Analysis of quetiapine in the patient's urine sample by gas chromatography–nitrogen–phosphorous detector (GC–NPD) determined that it contained 0.2 μg/mL of the parent compound (Table II) (17). Both the Microgenics® and Syva® immunoassays returned positive TCA results for the quetiapine dosed patient. This disagreement in the concentration cutoff for false positives occurring between native matrices and samples diluted in water is likely explained by the quetiapine metabolites present in patient specimens. Based on these data, it appears that quetiapine metabolites interfere with immunoassays for TCAs to a greater extent than the parent drug (17). Upon visual inspection of the structure of quetiapine and structures of common TCAs, one can recognize the structural similarities between them (Figure 1). This close structural relationship between quetiapine and the TCA drug class has been maintained as the probable cause for the observed interferences (17, 18).
False-Positive Results for TCA Immunoassays
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Quetiapine | Roche (Hitachi 911) (300 ng/mL cutoff of nortriptyline) | Microgenics® | 7.0 μg/mL | Spiking of dissolved tablets in H2O | (16) |
| Beckman LX20 | Microgenics Tricyclics Serum Tox EIA® | 10.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| Beckman LX20 | Microgenics Tricyclics Serum Tox EIA® | 0.2 μg/mL | Therapeutic dosing | (17) | |
| – | SYVA RapidTest d.a.u.® | 100.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| – | SYVA RapidTest d.a.u.® | 0.2 μg/mL | Therapeutic dosing | (17) | |
| – | Biosite Triage® | >1,000.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| – | Biosite Triage® | Did not occur | Therapeutic dosing | (17) | |
| – | Bio-Rad Tox/See® | Occurred | Case report | (18) |
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Quetiapine | Roche (Hitachi 911) (300 ng/mL cutoff of nortriptyline) | Microgenics® | 7.0 μg/mL | Spiking of dissolved tablets in H2O | (16) |
| Beckman LX20 | Microgenics Tricyclics Serum Tox EIA® | 10.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| Beckman LX20 | Microgenics Tricyclics Serum Tox EIA® | 0.2 μg/mL | Therapeutic dosing | (17) | |
| – | SYVA RapidTest d.a.u.® | 100.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| – | SYVA RapidTest d.a.u.® | 0.2 μg/mL | Therapeutic dosing | (17) | |
| – | Biosite Triage® | >1,000.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| – | Biosite Triage® | Did not occur | Therapeutic dosing | (17) | |
| – | Bio-Rad Tox/See® | Occurred | Case report | (18) |
False-Positive Results for TCA Immunoassays
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Quetiapine | Roche (Hitachi 911) (300 ng/mL cutoff of nortriptyline) | Microgenics® | 7.0 μg/mL | Spiking of dissolved tablets in H2O | (16) |
| Beckman LX20 | Microgenics Tricyclics Serum Tox EIA® | 10.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| Beckman LX20 | Microgenics Tricyclics Serum Tox EIA® | 0.2 μg/mL | Therapeutic dosing | (17) | |
| – | SYVA RapidTest d.a.u.® | 100.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| – | SYVA RapidTest d.a.u.® | 0.2 μg/mL | Therapeutic dosing | (17) | |
| – | Biosite Triage® | >1,000.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| – | Biosite Triage® | Did not occur | Therapeutic dosing | (17) | |
| – | Bio-Rad Tox/See® | Occurred | Case report | (18) |
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Quetiapine | Roche (Hitachi 911) (300 ng/mL cutoff of nortriptyline) | Microgenics® | 7.0 μg/mL | Spiking of dissolved tablets in H2O | (16) |
| Beckman LX20 | Microgenics Tricyclics Serum Tox EIA® | 10.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| Beckman LX20 | Microgenics Tricyclics Serum Tox EIA® | 0.2 μg/mL | Therapeutic dosing | (17) | |
| – | SYVA RapidTest d.a.u.® | 100.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| – | SYVA RapidTest d.a.u.® | 0.2 μg/mL | Therapeutic dosing | (17) | |
| – | Biosite Triage® | >1,000.0 μg/mL | Spiking of dissolved tablets in DFU | (17) | |
| – | Biosite Triage® | Did not occur | Therapeutic dosing | (17) | |
| – | Bio-Rad Tox/See® | Occurred | Case report | (18) |
Benzodiazepines
Benzodiazepines are widely prescribed as anxiolytics and hypnotics and are commonly detected in UDS. Immunoassay-based drug monitoring programs for benzodiazepines are well established; however, like most immunoassays, they are subject to false-positive results (Table III). Conversely, immunoassays for benzodiazepines also suffer from clinically significant false-negative results. The false negatives are not discussed here, but it is important that providers are aware of this limitation (21). Efavirenz (EFV) (6), used for the treatment of HIV, has been shown to cross-react with Biosite Triage 8® kit for benzodiazepines (Figure 1) (19). The authors created a blinded study in which half of the patients (n = 50) were therapeutically dosed with EFV and the other half (n = 50) were not. Running the Biosite Triage® kit for benzodiazepines, 46 of the 50 patients prescribed EFV returned a positive result, whereas 47 of the 50 patients not prescribed EFV were negative for benzodiazepines. Two other POC immunoassays (Drug Control 008® and Drug Screen Multi 5®) along with LC–MS-MS confirmation were then performed on the presumptive positive samples, all of which returned negative results (19). The authors also conducted spiking experiments with standards of EFV and its oxidized metabolite, 8-OH EFV, in DFU and revealed that both compounds are responsible for the observed false-positive results (Table III) (19). EFV, 8-OH EFV and many benzodiazepines are similar in structure, which may explain some of the observed cross-reactivity (Figure 1).
False-Positive Results for Benzodiazepine Immunoassays
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| EFV | – | Biosite Triage 8® | Occurred | Retrospective chart review | (19) |
| Sertraline | Abbot Architect and Abbot Aeroset® | Not mentioned | Occurred | Retrospective chart review | (20) |
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| EFV | – | Biosite Triage 8® | Occurred | Retrospective chart review | (19) |
| Sertraline | Abbot Architect and Abbot Aeroset® | Not mentioned | Occurred | Retrospective chart review | (20) |
False-Positive Results for Benzodiazepine Immunoassays
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| EFV | – | Biosite Triage 8® | Occurred | Retrospective chart review | (19) |
| Sertraline | Abbot Architect and Abbot Aeroset® | Not mentioned | Occurred | Retrospective chart review | (20) |
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay name . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| EFV | – | Biosite Triage 8® | Occurred | Retrospective chart review | (19) |
| Sertraline | Abbot Architect and Abbot Aeroset® | Not mentioned | Occurred | Retrospective chart review | (20) |
False-positive benzodiazepine results may also originate from the commonly prescribed antidepressant sertraline (20). A retrospective chart review was performed for all specimens in a 2-year span (January 2007–December 2008) that screened positive on the Abbott Architect® and Aeroset® platforms, but were negative by confirmatory testing (20). These false positives were then cross-referenced with the patients' prescription records. The authors concluded that 26.5% (26 samples) had false-positive benzodiazepine results related to sertraline use (Table III) although no spiking experiments were performed to support these observations (20).
Cannabinoids
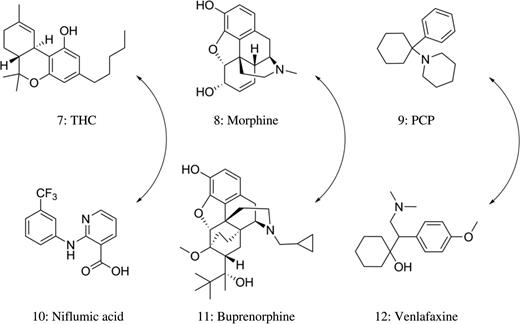
Structures of THC, morphine and PCP (top) along with cross-reacting compounds (bottom).
Patients prescribed antiretroviral therapy with EFV (6) produce urine samples that screened positive for THC exposure, despite the absence of 11-nor-Δ9-THC-9-COOH (Table IV). This interference has been attributed to EFVs major metabolite, EFV 8-glucuronide (EFV-8-G). The other major urinary metabolite, 8-OH EFV, and EFV were found to not interfere (22). Three immunoassays showed false-positive results for cannabinoid screening including Microgenics Corporation (Cedia® Dau Multi-Level THC), Biosite Incorporated (Triage® TOX Drug Screen) and Immunalysis Corporation [Cannabinoids (THCA/CTHC) Direct ELISA Kit] (22). Oosthuizen and Laurens (23) evaluated two POC devices (THC One Step Marijuana and Rapid Response Drugs-of-Abuse Test Strips) and two automated immunoassays (Roche Diagnostics Cannabinoids II and Beckman Coulter SYNCHRON Systems THC2) for THC false-positive results caused by EFV. They reported that the Rapid Response test strips yielded positive results in 28 of 30 patients taking 600 mg EFV therapy for at least 2 weeks, but the results from all of the other platforms were negative (Table IV) (23).
False-Positive Results for Cannabinoid Immunoassays
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Efavirenz | Cedia® Dau Multi-Level THC, Triage® TOX Drug Screen and Cannabinoids (THCA/CTHC) Direct ELISA Kit, cutoff 50 µg/L | Microgenics, BioSite and Immunalysis | Occurred | Case report | (22) |
| Rapid response drugs-of-abuse test strips, cutoff 50 ng/mL | BTNX | Occurred | Case report | (23) | |
| Ibuprofen | EMIT® d.a.u. cutoff 20 µg/L | Syva | Occurred | Case report | (24) |
| Naproxen | EMIT® d.a.u. cutoff 20 µg/L | Syva | Occurred | Case report | (24) |
| Niflumic acid | Kinetic interaction of microparticles in a solution (KIMS®), cutoff 50 ng/mL | Roche | 2.5 μg/mL | Spiking of standards in DFU | (25) |
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Efavirenz | Cedia® Dau Multi-Level THC, Triage® TOX Drug Screen and Cannabinoids (THCA/CTHC) Direct ELISA Kit, cutoff 50 µg/L | Microgenics, BioSite and Immunalysis | Occurred | Case report | (22) |
| Rapid response drugs-of-abuse test strips, cutoff 50 ng/mL | BTNX | Occurred | Case report | (23) | |
| Ibuprofen | EMIT® d.a.u. cutoff 20 µg/L | Syva | Occurred | Case report | (24) |
| Naproxen | EMIT® d.a.u. cutoff 20 µg/L | Syva | Occurred | Case report | (24) |
| Niflumic acid | Kinetic interaction of microparticles in a solution (KIMS®), cutoff 50 ng/mL | Roche | 2.5 μg/mL | Spiking of standards in DFU | (25) |
False-Positive Results for Cannabinoid Immunoassays
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Efavirenz | Cedia® Dau Multi-Level THC, Triage® TOX Drug Screen and Cannabinoids (THCA/CTHC) Direct ELISA Kit, cutoff 50 µg/L | Microgenics, BioSite and Immunalysis | Occurred | Case report | (22) |
| Rapid response drugs-of-abuse test strips, cutoff 50 ng/mL | BTNX | Occurred | Case report | (23) | |
| Ibuprofen | EMIT® d.a.u. cutoff 20 µg/L | Syva | Occurred | Case report | (24) |
| Naproxen | EMIT® d.a.u. cutoff 20 µg/L | Syva | Occurred | Case report | (24) |
| Niflumic acid | Kinetic interaction of microparticles in a solution (KIMS®), cutoff 50 ng/mL | Roche | 2.5 μg/mL | Spiking of standards in DFU | (25) |
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Efavirenz | Cedia® Dau Multi-Level THC, Triage® TOX Drug Screen and Cannabinoids (THCA/CTHC) Direct ELISA Kit, cutoff 50 µg/L | Microgenics, BioSite and Immunalysis | Occurred | Case report | (22) |
| Rapid response drugs-of-abuse test strips, cutoff 50 ng/mL | BTNX | Occurred | Case report | (23) | |
| Ibuprofen | EMIT® d.a.u. cutoff 20 µg/L | Syva | Occurred | Case report | (24) |
| Naproxen | EMIT® d.a.u. cutoff 20 µg/L | Syva | Occurred | Case report | (24) |
| Niflumic acid | Kinetic interaction of microparticles in a solution (KIMS®), cutoff 50 ng/mL | Roche | 2.5 μg/mL | Spiking of standards in DFU | (25) |
Niflumic acid (10) is a nonsteroidal, anti-inflammatory drug that inhibits cyclooxygenase-2 (Figure 2). A patient dosing study revealed that all 55 urine samples from five volunteers taking niflumic acid returned a positive THC result when analyzed by the KIMS® assay but were negative when analyzed by the EMIT THC® assay (25). Niflumic acid standards were added to DFU at 13 concentrations ranging from 1.25 to 1,000 μg/mL. A concentration of niflumic acid at ∼2.5 μg/mL had a KIMS® response equal to the 50 ng/mL cutoff (Table IV) (25).
Cotten et al. (26) identified several soap-based products that could potentially cause a false positive with the Vitros THC® (Cannabinoid) immunoassay on the Vitros 5600 (Ortho Clinical Diagnostics, Inc., Rochester, NY, USA). Four commercial baby soaps caused assay interference sufficient to yield a positive screen result (cutoff 20 ng/mL) (26). Despite the identification of several surfactant components contributing to the interference, the authors maintain that the exact mechanism for the false-positive results remains unclear (26).
Opiates
Opiates belong to a large class of compounds characterized by their ability to interact with endogenous opiate receptors (27). Opiate immunoassays typically target morphine (8) (Figure 2) and codeine, both naturally isolated from the opium poppy (Papaver somniferum). Semisynthetic opiates are similar in structure to morphine, whereas synthetic opioids that bind to opiate receptors generally require separate immunoassays for screening purposes.
Naloxone cross-reactivity with opiate immunoassays has been reported. Urine samples supplemented with 6.1 µg/mL of naloxone or greater were ‘opiate positive’ with the opiate CEDIA® assay (28). This finding is of particular importance as most patients with a suspected opiate overdose are treated with naloxone (Table V) (28). Buprenorphine (11) is a semisynthetic opioid (Figure 2) commonly administered in opiate agonist therapy to manage the patients dependent on opioids (48). When the Microgenics buprenorphine CEDIA® assay was evaluated for cross-reactivity, Pavlic et al. (30) demonstrated that 0.12 mg/mL morphine, 0.32 mg/mL methadone, 0.03 mg/mL codeine, 0.06 mg/mL dihydrocodeine and 0.52 mg/mL morphine-3-glucuronide produced positive results. This observed cross-reactivity is not unexpected as the structural characteristics of buprenorphine are similar to natural opiate derivatives (Table V). Patients are frequently tested simultaneously for buprenorphine compliance and avoidance of illicit opioids as a condition of continuance with opioid substitution therapy. It is particularly troublesome that a false-positive buprenorphine screening test could be caused by heroin use, which potentially yields large quantities of morphine in urine.
False-Positive Results for Opiate Immunoassays
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Amisulpride and sulpiride | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Thermo Fisher Scientific | 130 mg/L for amisulpride and 250 mg/L for sulpiride | Spiking of standards in DFU | (29) |
| Codeine, dihydrocodeine, morphine, methadone and morphine-3-glucuronide | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Microgenics | 30 mg/L (codeine), 60 mg/L (dihydrocodeine), 120 mg/L (morphine), 320 mg/L (methadone) and 520 mg/L (morphine-3-glucuronide) | Spiking of standards in DFU | (30) |
| Creatinine | EMIT® d.a.u. methadone, cutoff 300 ng/mL | Syva | Occurred | Case report | (31) |
| Dihydrocodeine | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Microgenics | Occurred | Case report | (32) |
| Diphenhydramine | Lateral-flow immunoassay for methadone, cutoff 300 ng/mL | One Step Multi-Drug Screen (ACON Laboratories) | 100 μg/mL | Spiking of standards in DFU | (33) |
| EMIT II® propoxyphene, cutoff 300 ng/mL | Syva | 200 μg/L | Spiking of standards in DFU | (34) | |
| Levofloxacine, ofloxacine | AxSYM fluorescence polarization immunoassay morphine, cutoff 300 ng/mL | Abbott | 1,700 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, enoxacine | CEDIA morphine®, cutoff 300 ng/mL | Microgenics | 60–1,700 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, enoxacine, gatifloxacine | EMIT II® morphine, cutoff 300 ng/mL | Syva | 140–600 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, lomefloxacine, moxifloxacine, ciprofloxacine, norfloxacine | Abuscreen OnLine morphine, cutoff 300 ng/mL | Roche | 200–1,700 μg/mL | Spiking of standards in DFU | (35) |
| Morphine | CEDIA buprenorphine assay®, cutoff 10 ng/mL | Microgenics | Occurred | Case report | (36) |
| Naloxone | CEDIA opiate®, cutoff 300 ng/mL | Microgenics | 6,100 μg/L | Spiking of standards in DFU | (28) |
| Ofloxacin | Fluorescence immunoassay for opiates | Triage TM and Multi-5M | Occurred | Case report | (37) |
| EMIT II® opiates, cutoff 300 µg/L | Syva | 200 mg/L | Spiking of standards in DFU | (38) | |
| Pentazocine | Cedia heroin metabolite (6-AM) assay cutoff 10 ng/mL | Microgenics | Occurred | Case report | (39) |
| Psychotropic drugs | Kinetic interaction of microparticles in a solution (KIMS®) methadone, cutoff 300 ng/mL | Roche Integra 800 | 8 mg/L cyamemazine, 57 mg/L alimemazine, 5 mg/L levomepromazine, 20 mg/L chlorpromazine, 100 mg/L clomipramine and 100 mg/L thioridazine | Spiking of standards in DFU | (40) |
| Quetiapine | Kinetic interaction of microparticles in solution (KIMS®) methadone cutoff 300 ng/mL | Roche COBAS Integra Methadone II test kit | Occurred | Case report | (41–43) |
| Rifampicin | KIMS® opiates (kinetic interaction of microparticles in solution) | Cobas Integra | 156–5,000 µg/L | Spiking of standards in DFU | (44) |
| Tapentadol | DRI methadone enzyme immunoassay cutoff 130 ng/mL | Microgenics | 6,500 ng/mL | Spiking of standards in DFU | (45) |
| Tramadol | CEDIA buprenorphine assay® cutoff of 5 ng/mL | Microgenics | 100 mg/L | Standard solutions of tramadol | (46) |
| Lateral-flow immunochrom buprenorphine assay, cutoff 5 ng/mL | Rapid One Buprenorphine Test Cards (Am. Bio Medica) | 50 mg/L | Standard solutions of tramadol | (46) | |
| Lateral-flow immunochromatographic buprenorphine assay, cutoff 5 ng/mL | QuikStrip OneStep Buprenorphine Test strips (CLIAwaived) | Occurred | Case report | (46) | |
| Verapamil | Methadone, cutoff 300 ng/mL | Diagnostic Reagents, Inc. (DRI) | 20 mg/L | Spiking of standards in DFU | (47) |
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Amisulpride and sulpiride | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Thermo Fisher Scientific | 130 mg/L for amisulpride and 250 mg/L for sulpiride | Spiking of standards in DFU | (29) |
| Codeine, dihydrocodeine, morphine, methadone and morphine-3-glucuronide | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Microgenics | 30 mg/L (codeine), 60 mg/L (dihydrocodeine), 120 mg/L (morphine), 320 mg/L (methadone) and 520 mg/L (morphine-3-glucuronide) | Spiking of standards in DFU | (30) |
| Creatinine | EMIT® d.a.u. methadone, cutoff 300 ng/mL | Syva | Occurred | Case report | (31) |
| Dihydrocodeine | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Microgenics | Occurred | Case report | (32) |
| Diphenhydramine | Lateral-flow immunoassay for methadone, cutoff 300 ng/mL | One Step Multi-Drug Screen (ACON Laboratories) | 100 μg/mL | Spiking of standards in DFU | (33) |
| EMIT II® propoxyphene, cutoff 300 ng/mL | Syva | 200 μg/L | Spiking of standards in DFU | (34) | |
| Levofloxacine, ofloxacine | AxSYM fluorescence polarization immunoassay morphine, cutoff 300 ng/mL | Abbott | 1,700 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, enoxacine | CEDIA morphine®, cutoff 300 ng/mL | Microgenics | 60–1,700 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, enoxacine, gatifloxacine | EMIT II® morphine, cutoff 300 ng/mL | Syva | 140–600 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, lomefloxacine, moxifloxacine, ciprofloxacine, norfloxacine | Abuscreen OnLine morphine, cutoff 300 ng/mL | Roche | 200–1,700 μg/mL | Spiking of standards in DFU | (35) |
| Morphine | CEDIA buprenorphine assay®, cutoff 10 ng/mL | Microgenics | Occurred | Case report | (36) |
| Naloxone | CEDIA opiate®, cutoff 300 ng/mL | Microgenics | 6,100 μg/L | Spiking of standards in DFU | (28) |
| Ofloxacin | Fluorescence immunoassay for opiates | Triage TM and Multi-5M | Occurred | Case report | (37) |
| EMIT II® opiates, cutoff 300 µg/L | Syva | 200 mg/L | Spiking of standards in DFU | (38) | |
| Pentazocine | Cedia heroin metabolite (6-AM) assay cutoff 10 ng/mL | Microgenics | Occurred | Case report | (39) |
| Psychotropic drugs | Kinetic interaction of microparticles in a solution (KIMS®) methadone, cutoff 300 ng/mL | Roche Integra 800 | 8 mg/L cyamemazine, 57 mg/L alimemazine, 5 mg/L levomepromazine, 20 mg/L chlorpromazine, 100 mg/L clomipramine and 100 mg/L thioridazine | Spiking of standards in DFU | (40) |
| Quetiapine | Kinetic interaction of microparticles in solution (KIMS®) methadone cutoff 300 ng/mL | Roche COBAS Integra Methadone II test kit | Occurred | Case report | (41–43) |
| Rifampicin | KIMS® opiates (kinetic interaction of microparticles in solution) | Cobas Integra | 156–5,000 µg/L | Spiking of standards in DFU | (44) |
| Tapentadol | DRI methadone enzyme immunoassay cutoff 130 ng/mL | Microgenics | 6,500 ng/mL | Spiking of standards in DFU | (45) |
| Tramadol | CEDIA buprenorphine assay® cutoff of 5 ng/mL | Microgenics | 100 mg/L | Standard solutions of tramadol | (46) |
| Lateral-flow immunochrom buprenorphine assay, cutoff 5 ng/mL | Rapid One Buprenorphine Test Cards (Am. Bio Medica) | 50 mg/L | Standard solutions of tramadol | (46) | |
| Lateral-flow immunochromatographic buprenorphine assay, cutoff 5 ng/mL | QuikStrip OneStep Buprenorphine Test strips (CLIAwaived) | Occurred | Case report | (46) | |
| Verapamil | Methadone, cutoff 300 ng/mL | Diagnostic Reagents, Inc. (DRI) | 20 mg/L | Spiking of standards in DFU | (47) |
False-Positive Results for Opiate Immunoassays
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Amisulpride and sulpiride | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Thermo Fisher Scientific | 130 mg/L for amisulpride and 250 mg/L for sulpiride | Spiking of standards in DFU | (29) |
| Codeine, dihydrocodeine, morphine, methadone and morphine-3-glucuronide | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Microgenics | 30 mg/L (codeine), 60 mg/L (dihydrocodeine), 120 mg/L (morphine), 320 mg/L (methadone) and 520 mg/L (morphine-3-glucuronide) | Spiking of standards in DFU | (30) |
| Creatinine | EMIT® d.a.u. methadone, cutoff 300 ng/mL | Syva | Occurred | Case report | (31) |
| Dihydrocodeine | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Microgenics | Occurred | Case report | (32) |
| Diphenhydramine | Lateral-flow immunoassay for methadone, cutoff 300 ng/mL | One Step Multi-Drug Screen (ACON Laboratories) | 100 μg/mL | Spiking of standards in DFU | (33) |
| EMIT II® propoxyphene, cutoff 300 ng/mL | Syva | 200 μg/L | Spiking of standards in DFU | (34) | |
| Levofloxacine, ofloxacine | AxSYM fluorescence polarization immunoassay morphine, cutoff 300 ng/mL | Abbott | 1,700 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, enoxacine | CEDIA morphine®, cutoff 300 ng/mL | Microgenics | 60–1,700 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, enoxacine, gatifloxacine | EMIT II® morphine, cutoff 300 ng/mL | Syva | 140–600 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, lomefloxacine, moxifloxacine, ciprofloxacine, norfloxacine | Abuscreen OnLine morphine, cutoff 300 ng/mL | Roche | 200–1,700 μg/mL | Spiking of standards in DFU | (35) |
| Morphine | CEDIA buprenorphine assay®, cutoff 10 ng/mL | Microgenics | Occurred | Case report | (36) |
| Naloxone | CEDIA opiate®, cutoff 300 ng/mL | Microgenics | 6,100 μg/L | Spiking of standards in DFU | (28) |
| Ofloxacin | Fluorescence immunoassay for opiates | Triage TM and Multi-5M | Occurred | Case report | (37) |
| EMIT II® opiates, cutoff 300 µg/L | Syva | 200 mg/L | Spiking of standards in DFU | (38) | |
| Pentazocine | Cedia heroin metabolite (6-AM) assay cutoff 10 ng/mL | Microgenics | Occurred | Case report | (39) |
| Psychotropic drugs | Kinetic interaction of microparticles in a solution (KIMS®) methadone, cutoff 300 ng/mL | Roche Integra 800 | 8 mg/L cyamemazine, 57 mg/L alimemazine, 5 mg/L levomepromazine, 20 mg/L chlorpromazine, 100 mg/L clomipramine and 100 mg/L thioridazine | Spiking of standards in DFU | (40) |
| Quetiapine | Kinetic interaction of microparticles in solution (KIMS®) methadone cutoff 300 ng/mL | Roche COBAS Integra Methadone II test kit | Occurred | Case report | (41–43) |
| Rifampicin | KIMS® opiates (kinetic interaction of microparticles in solution) | Cobas Integra | 156–5,000 µg/L | Spiking of standards in DFU | (44) |
| Tapentadol | DRI methadone enzyme immunoassay cutoff 130 ng/mL | Microgenics | 6,500 ng/mL | Spiking of standards in DFU | (45) |
| Tramadol | CEDIA buprenorphine assay® cutoff of 5 ng/mL | Microgenics | 100 mg/L | Standard solutions of tramadol | (46) |
| Lateral-flow immunochrom buprenorphine assay, cutoff 5 ng/mL | Rapid One Buprenorphine Test Cards (Am. Bio Medica) | 50 mg/L | Standard solutions of tramadol | (46) | |
| Lateral-flow immunochromatographic buprenorphine assay, cutoff 5 ng/mL | QuikStrip OneStep Buprenorphine Test strips (CLIAwaived) | Occurred | Case report | (46) | |
| Verapamil | Methadone, cutoff 300 ng/mL | Diagnostic Reagents, Inc. (DRI) | 20 mg/L | Spiking of standards in DFU | (47) |
| Cross-reacting drug . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Amisulpride and sulpiride | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Thermo Fisher Scientific | 130 mg/L for amisulpride and 250 mg/L for sulpiride | Spiking of standards in DFU | (29) |
| Codeine, dihydrocodeine, morphine, methadone and morphine-3-glucuronide | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Microgenics | 30 mg/L (codeine), 60 mg/L (dihydrocodeine), 120 mg/L (morphine), 320 mg/L (methadone) and 520 mg/L (morphine-3-glucuronide) | Spiking of standards in DFU | (30) |
| Creatinine | EMIT® d.a.u. methadone, cutoff 300 ng/mL | Syva | Occurred | Case report | (31) |
| Dihydrocodeine | CEDIA buprenorphine assay®, cutoff 5 ng/mL | Microgenics | Occurred | Case report | (32) |
| Diphenhydramine | Lateral-flow immunoassay for methadone, cutoff 300 ng/mL | One Step Multi-Drug Screen (ACON Laboratories) | 100 μg/mL | Spiking of standards in DFU | (33) |
| EMIT II® propoxyphene, cutoff 300 ng/mL | Syva | 200 μg/L | Spiking of standards in DFU | (34) | |
| Levofloxacine, ofloxacine | AxSYM fluorescence polarization immunoassay morphine, cutoff 300 ng/mL | Abbott | 1,700 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, enoxacine | CEDIA morphine®, cutoff 300 ng/mL | Microgenics | 60–1,700 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, enoxacine, gatifloxacine | EMIT II® morphine, cutoff 300 ng/mL | Syva | 140–600 μg/mL | Spiking of standards in DFU | (35) |
| Levofloxacine, ofloxacine, pefloxacine, lomefloxacine, moxifloxacine, ciprofloxacine, norfloxacine | Abuscreen OnLine morphine, cutoff 300 ng/mL | Roche | 200–1,700 μg/mL | Spiking of standards in DFU | (35) |
| Morphine | CEDIA buprenorphine assay®, cutoff 10 ng/mL | Microgenics | Occurred | Case report | (36) |
| Naloxone | CEDIA opiate®, cutoff 300 ng/mL | Microgenics | 6,100 μg/L | Spiking of standards in DFU | (28) |
| Ofloxacin | Fluorescence immunoassay for opiates | Triage TM and Multi-5M | Occurred | Case report | (37) |
| EMIT II® opiates, cutoff 300 µg/L | Syva | 200 mg/L | Spiking of standards in DFU | (38) | |
| Pentazocine | Cedia heroin metabolite (6-AM) assay cutoff 10 ng/mL | Microgenics | Occurred | Case report | (39) |
| Psychotropic drugs | Kinetic interaction of microparticles in a solution (KIMS®) methadone, cutoff 300 ng/mL | Roche Integra 800 | 8 mg/L cyamemazine, 57 mg/L alimemazine, 5 mg/L levomepromazine, 20 mg/L chlorpromazine, 100 mg/L clomipramine and 100 mg/L thioridazine | Spiking of standards in DFU | (40) |
| Quetiapine | Kinetic interaction of microparticles in solution (KIMS®) methadone cutoff 300 ng/mL | Roche COBAS Integra Methadone II test kit | Occurred | Case report | (41–43) |
| Rifampicin | KIMS® opiates (kinetic interaction of microparticles in solution) | Cobas Integra | 156–5,000 µg/L | Spiking of standards in DFU | (44) |
| Tapentadol | DRI methadone enzyme immunoassay cutoff 130 ng/mL | Microgenics | 6,500 ng/mL | Spiking of standards in DFU | (45) |
| Tramadol | CEDIA buprenorphine assay® cutoff of 5 ng/mL | Microgenics | 100 mg/L | Standard solutions of tramadol | (46) |
| Lateral-flow immunochrom buprenorphine assay, cutoff 5 ng/mL | Rapid One Buprenorphine Test Cards (Am. Bio Medica) | 50 mg/L | Standard solutions of tramadol | (46) | |
| Lateral-flow immunochromatographic buprenorphine assay, cutoff 5 ng/mL | QuikStrip OneStep Buprenorphine Test strips (CLIAwaived) | Occurred | Case report | (46) | |
| Verapamil | Methadone, cutoff 300 ng/mL | Diagnostic Reagents, Inc. (DRI) | 20 mg/L | Spiking of standards in DFU | (47) |
The buprenorphine assay is also subject to cross-reactivity from the synthetic opioid tramadol (Table V) (46). Tramadol interference with the CEDIA® buprenorphine assay is clinically relevant if a 5 ng/mL calibration cutoff is used. However, when the cutoff was increased from 5 to 20 ng/mL, all false positives were eliminated (Table V) (46). Amisulpride and sulpiride were also found to cross-react with the CEDIA® buprenorphine assay. Although the cross-reactivity is low, it remains significant given the high concentrations of these compounds that are commonly found in urine relative to the low cutoff (5 μg/L) of the buprenorphine immunoassay (29).
Methadone, a synthetic opioid, is used medically as an analgesic for the management of severe pain and in maintenance doses for patients with opioid dependency. Several reports exist regarding false-positive results for methadone screening for patients prescribed quetiapine (41, 42). Cherwinski et al. (49) reported that 125 mg/day doses of quetiapine were sufficient to produce a false positive of methadone screening results. Quetiapine is a widely used antipsychotic and is structurally similar to methadone and is maintained as the probable cause for the observed cross-reactivity (Table V) (43).
Miscellaneous drugs
This section is a compilation of drugs of abuse that do not fit into a particular class and are discussed individually. Phencyclidine, commonly known as PCP (9), is a synthetic, dissociative anesthetic, which has been sporadically abused for the last 50 years (Figure 2) (50). Many immunoassays that are available purport to specifically identify PCP in urine specimens. However, the low prevalence of PCP use combined with the low specificity of PCP immunoassays makes the positive predictive value of PCP screen very low (Table VI).
False-Positive Results for Miscellaneous Drug (PCP, LSD and Barbiturates) Immunoassays
| . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Cross-reacting drug for PCP | |||||
| Lamotrigine | – | Bio-Rad Tox/See® | 7.6 μg/mL (serum) | Case report | (50) |
| MDPV | Beckman Coulter DxC 800 ®, cutoff 25 ng/mL | Synchron® | 3.1 μg/mL | Spiking of standards in DFU | (51) |
| Tramadol | Not defined | Syva EMIT II Plus® | 5.4 μg/mL | Case report | (52) |
| Siemens ADVIA 1800® | DRI PCP Microgenics®, cutoff 25 ng/mL | 500 μg/mL at lowest cutoff 25 ng/mL | Spiking of standards in DFU | (53) | |
| Not defined | Syva EMIT II® | Occurred | Case report | (54) | |
| Siemens Dimension® | Syva EMIT II® | Occurred | Retrospective chart review | (3) | |
| Venlafaxine | – | Syva RapidTest d.a.u.®, cutoff 25 ng/mL | 200 μg/mL | Spiking of standards in DFU | (55) |
| Abbot AxSYM® | FPIA for PCP | Occurred cutoff 25 ng/mL | Case report | (56) | |
| – | Instant-View Multi-Drug Screen® | Occurred | Case report | (57) | |
| Cross-reacting drug for LSD | |||||
| Ambroxol | CEDIA DAU LSD assay cutoff 0.5 ng/mL | Hitachi 917-analyzer | 3 mg/L | Spiking of standards in DFU | (58) |
| Amitriptyline, benzphetamine, bupropion, buspirone, cephradine, chlorpromazine, desipramine, diltiazem, doxepin, fentanyl, fluoxetine, haloperidol, imipramine, labetalol, metoclopramide, prochlorperazine, risperidone, sertraline, thioridazine, trazodone and verapamil | Emit II LSD® assay from Behring diagnostics cutoff 0.5 ng/mL | Syva-30R automated assay | Variable (0.1–<1,000 mg/L) | Spiking of standards in DFU | (59) |
| Ergonovine, lysergol, brompheniramine maleate, imipramine HCl and methylphenidate HCl | EMIT II LSD® cutoff 500 pg/mL | Dade Behring | 4,000 ng/mL (ergonovine), 25,000 ng/mL (lysergol and brompheniramine maleate), 50,000 ng/mL (imipramine HCl) and 50,000 ng/mL (methylphenidate HCl) | Spiking of standards in DFU | (60) |
| Fentanyl | CEDIA LSD® assay and Emit II LSD® assay | Microgenics and SYVA | 40 μg/L | Spiking of standards in DFU | (61) |
| Sertraline | CEDIA® | Microgenics | 1.5 mg/L | Spiking of standards in DFU | (62) |
| Cross-reacting drug for barbiturates | |||||
| Ibuprofen | FPIA (cutoff 500 μg/L) | Abbott TDx | Occurred | Case report | (24) |
| Naproxen | FPIA (cutoff 500 μg/L) | Abbott TDx | Occurred | Case report | (24) |
| . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Cross-reacting drug for PCP | |||||
| Lamotrigine | – | Bio-Rad Tox/See® | 7.6 μg/mL (serum) | Case report | (50) |
| MDPV | Beckman Coulter DxC 800 ®, cutoff 25 ng/mL | Synchron® | 3.1 μg/mL | Spiking of standards in DFU | (51) |
| Tramadol | Not defined | Syva EMIT II Plus® | 5.4 μg/mL | Case report | (52) |
| Siemens ADVIA 1800® | DRI PCP Microgenics®, cutoff 25 ng/mL | 500 μg/mL at lowest cutoff 25 ng/mL | Spiking of standards in DFU | (53) | |
| Not defined | Syva EMIT II® | Occurred | Case report | (54) | |
| Siemens Dimension® | Syva EMIT II® | Occurred | Retrospective chart review | (3) | |
| Venlafaxine | – | Syva RapidTest d.a.u.®, cutoff 25 ng/mL | 200 μg/mL | Spiking of standards in DFU | (55) |
| Abbot AxSYM® | FPIA for PCP | Occurred cutoff 25 ng/mL | Case report | (56) | |
| – | Instant-View Multi-Drug Screen® | Occurred | Case report | (57) | |
| Cross-reacting drug for LSD | |||||
| Ambroxol | CEDIA DAU LSD assay cutoff 0.5 ng/mL | Hitachi 917-analyzer | 3 mg/L | Spiking of standards in DFU | (58) |
| Amitriptyline, benzphetamine, bupropion, buspirone, cephradine, chlorpromazine, desipramine, diltiazem, doxepin, fentanyl, fluoxetine, haloperidol, imipramine, labetalol, metoclopramide, prochlorperazine, risperidone, sertraline, thioridazine, trazodone and verapamil | Emit II LSD® assay from Behring diagnostics cutoff 0.5 ng/mL | Syva-30R automated assay | Variable (0.1–<1,000 mg/L) | Spiking of standards in DFU | (59) |
| Ergonovine, lysergol, brompheniramine maleate, imipramine HCl and methylphenidate HCl | EMIT II LSD® cutoff 500 pg/mL | Dade Behring | 4,000 ng/mL (ergonovine), 25,000 ng/mL (lysergol and brompheniramine maleate), 50,000 ng/mL (imipramine HCl) and 50,000 ng/mL (methylphenidate HCl) | Spiking of standards in DFU | (60) |
| Fentanyl | CEDIA LSD® assay and Emit II LSD® assay | Microgenics and SYVA | 40 μg/L | Spiking of standards in DFU | (61) |
| Sertraline | CEDIA® | Microgenics | 1.5 mg/L | Spiking of standards in DFU | (62) |
| Cross-reacting drug for barbiturates | |||||
| Ibuprofen | FPIA (cutoff 500 μg/L) | Abbott TDx | Occurred | Case report | (24) |
| Naproxen | FPIA (cutoff 500 μg/L) | Abbott TDx | Occurred | Case report | (24) |
False-Positive Results for Miscellaneous Drug (PCP, LSD and Barbiturates) Immunoassays
| . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Cross-reacting drug for PCP | |||||
| Lamotrigine | – | Bio-Rad Tox/See® | 7.6 μg/mL (serum) | Case report | (50) |
| MDPV | Beckman Coulter DxC 800 ®, cutoff 25 ng/mL | Synchron® | 3.1 μg/mL | Spiking of standards in DFU | (51) |
| Tramadol | Not defined | Syva EMIT II Plus® | 5.4 μg/mL | Case report | (52) |
| Siemens ADVIA 1800® | DRI PCP Microgenics®, cutoff 25 ng/mL | 500 μg/mL at lowest cutoff 25 ng/mL | Spiking of standards in DFU | (53) | |
| Not defined | Syva EMIT II® | Occurred | Case report | (54) | |
| Siemens Dimension® | Syva EMIT II® | Occurred | Retrospective chart review | (3) | |
| Venlafaxine | – | Syva RapidTest d.a.u.®, cutoff 25 ng/mL | 200 μg/mL | Spiking of standards in DFU | (55) |
| Abbot AxSYM® | FPIA for PCP | Occurred cutoff 25 ng/mL | Case report | (56) | |
| – | Instant-View Multi-Drug Screen® | Occurred | Case report | (57) | |
| Cross-reacting drug for LSD | |||||
| Ambroxol | CEDIA DAU LSD assay cutoff 0.5 ng/mL | Hitachi 917-analyzer | 3 mg/L | Spiking of standards in DFU | (58) |
| Amitriptyline, benzphetamine, bupropion, buspirone, cephradine, chlorpromazine, desipramine, diltiazem, doxepin, fentanyl, fluoxetine, haloperidol, imipramine, labetalol, metoclopramide, prochlorperazine, risperidone, sertraline, thioridazine, trazodone and verapamil | Emit II LSD® assay from Behring diagnostics cutoff 0.5 ng/mL | Syva-30R automated assay | Variable (0.1–<1,000 mg/L) | Spiking of standards in DFU | (59) |
| Ergonovine, lysergol, brompheniramine maleate, imipramine HCl and methylphenidate HCl | EMIT II LSD® cutoff 500 pg/mL | Dade Behring | 4,000 ng/mL (ergonovine), 25,000 ng/mL (lysergol and brompheniramine maleate), 50,000 ng/mL (imipramine HCl) and 50,000 ng/mL (methylphenidate HCl) | Spiking of standards in DFU | (60) |
| Fentanyl | CEDIA LSD® assay and Emit II LSD® assay | Microgenics and SYVA | 40 μg/L | Spiking of standards in DFU | (61) |
| Sertraline | CEDIA® | Microgenics | 1.5 mg/L | Spiking of standards in DFU | (62) |
| Cross-reacting drug for barbiturates | |||||
| Ibuprofen | FPIA (cutoff 500 μg/L) | Abbott TDx | Occurred | Case report | (24) |
| Naproxen | FPIA (cutoff 500 μg/L) | Abbott TDx | Occurred | Case report | (24) |
| . | Immunoassay platform (positive cutoff) . | Immunoassay manufacturer . | Level which false (+) occurred . | Level of evidence . | Reference . |
|---|---|---|---|---|---|
| Cross-reacting drug for PCP | |||||
| Lamotrigine | – | Bio-Rad Tox/See® | 7.6 μg/mL (serum) | Case report | (50) |
| MDPV | Beckman Coulter DxC 800 ®, cutoff 25 ng/mL | Synchron® | 3.1 μg/mL | Spiking of standards in DFU | (51) |
| Tramadol | Not defined | Syva EMIT II Plus® | 5.4 μg/mL | Case report | (52) |
| Siemens ADVIA 1800® | DRI PCP Microgenics®, cutoff 25 ng/mL | 500 μg/mL at lowest cutoff 25 ng/mL | Spiking of standards in DFU | (53) | |
| Not defined | Syva EMIT II® | Occurred | Case report | (54) | |
| Siemens Dimension® | Syva EMIT II® | Occurred | Retrospective chart review | (3) | |
| Venlafaxine | – | Syva RapidTest d.a.u.®, cutoff 25 ng/mL | 200 μg/mL | Spiking of standards in DFU | (55) |
| Abbot AxSYM® | FPIA for PCP | Occurred cutoff 25 ng/mL | Case report | (56) | |
| – | Instant-View Multi-Drug Screen® | Occurred | Case report | (57) | |
| Cross-reacting drug for LSD | |||||
| Ambroxol | CEDIA DAU LSD assay cutoff 0.5 ng/mL | Hitachi 917-analyzer | 3 mg/L | Spiking of standards in DFU | (58) |
| Amitriptyline, benzphetamine, bupropion, buspirone, cephradine, chlorpromazine, desipramine, diltiazem, doxepin, fentanyl, fluoxetine, haloperidol, imipramine, labetalol, metoclopramide, prochlorperazine, risperidone, sertraline, thioridazine, trazodone and verapamil | Emit II LSD® assay from Behring diagnostics cutoff 0.5 ng/mL | Syva-30R automated assay | Variable (0.1–<1,000 mg/L) | Spiking of standards in DFU | (59) |
| Ergonovine, lysergol, brompheniramine maleate, imipramine HCl and methylphenidate HCl | EMIT II LSD® cutoff 500 pg/mL | Dade Behring | 4,000 ng/mL (ergonovine), 25,000 ng/mL (lysergol and brompheniramine maleate), 50,000 ng/mL (imipramine HCl) and 50,000 ng/mL (methylphenidate HCl) | Spiking of standards in DFU | (60) |
| Fentanyl | CEDIA LSD® assay and Emit II LSD® assay | Microgenics and SYVA | 40 μg/L | Spiking of standards in DFU | (61) |
| Sertraline | CEDIA® | Microgenics | 1.5 mg/L | Spiking of standards in DFU | (62) |
| Cross-reacting drug for barbiturates | |||||
| Ibuprofen | FPIA (cutoff 500 μg/L) | Abbott TDx | Occurred | Case report | (24) |
| Naproxen | FPIA (cutoff 500 μg/L) | Abbott TDx | Occurred | Case report | (24) |
Venlafaxine (12), often prescribed as an antidepressant, has been shown to cause false-positive PCP results with multiple immunoassays (Figure 2). One case report presented by Bond et al. (56) describes a false-positive PCP immunoassay from a patient who overdosed on venlafaxine. A UDS performed using the Abbott AxSYM® platform was positive for PCP. Confirmative testing via GC–MS for PCP was negative. The only substance identified in the patient's serum via GC–MS was venlafaxine that was present at a concentration of >24 μg/mL, a concentration almost 1,000 times the therapeutic dose. The authors spiked DFU with an equivalent quantity of venlafaxine and its metabolite O-desmethylvenlafaxine and analyzed the samples using the Abbott AxSYM® platform. The spiked samples were positive for PCP even when diluted four times with DFU (56). Another instance of venlafaxine inference for PCP comes from a case report by Sena et al. Three separate patient urine specimens tested with the Syva RapidTest d.a.u.® method were positive for PCP using a cutoff of 25-ng/mL (Table VI) (55). Venlafaxine was found to be common in all three patients. The three samples were then retested on different immunoassay platforms for PCP. These included Syva Emit II Plus® PCP assay on the Roche Cobas c501® platform, the OnTrak TesTstik PCP® kit and the Biosite Triage 8®, all of which had a cutoff of 25 ng/mL and all of which returned negative results for PCP. GC–MS testing confirmed these negative PCP results for all three patients (Table VI) (55).
Tramadol also causes false-positive PCP immunoassay results. One study reports two cases of false-positive PCP urine screens from patients taking tramadol using the DRI PCP Microgenics® kit on the Siemens ADVIA 1800® platform (53). No PCP was detected in these specimens with GC–MS. The first patient had acknowledged to taking 10 times her prescribed tramadol dose each day for the 2 days prior to her urine samples being collected. A broad-spectrum GC–MS analysis was conducted on the patient's urine sample that showed a large and distorted peak for tramadol, indicating a high but undefined concentration of the drug. The second patient had also consumed a large quantity of tramadol (eighty-four 50 mg tramadol tablets) and showed similar GC–MS results. The authors performed dilution studies of tramadol, O-desmethyltramadol and N-desmethyltramadol standards in DFU and demonstrated that concentrations of 500 μg/mL for both tramadol and N-desmethyltramadol were found to trigger a positive PCP response at a cutoff of 25 ng/mL (53). False positives for PCP from tramadol overdose were also noted in two patients using the Syva EMIT II Plus® immunoassay (Table VI) (54). Both patients were admitted to the emergency department with PCP positive UDSs. Confirmatory analysis by GC–MS showed no detectable PCP. Tramadol, however, was found in high amounts although exact quantification was not established (54). The authors suggest that these PCP false positives are due to the two compounds' structural similarities (54).
3,4-Methylenedioxypyrovalerone (MDPV) is a synthetic cathinone found in illicit ‘bath salts’ and was revealed by Macher and Penders to cause false-positive PCP results. The case involved a patient who was admitted to the emergency department with an apparent overdose. A urine sample was collected and analyzed with a Beckman Coulter DxC 800® method run on a Synchron® platform (51). Confirmatory testing was not conducted on the patient's urine sample but was accomplished via GC–MS using a postmortem blood sample. The results indicated no PCP was present in the patient's blood at the time of death. MDPV, however, was detected in the blood at a concentration of 1.0 μg/mL, prompting further investigative studies (Table VI). Dilutions of MDPV standards in DFU were made and analyzed, which showed that an MDPV concentration of >3.1 μg/mL can cause false-positive results for PCP (51).
Immunoassays for the hallucinogen, lysergic acid diethylamide (LSD), have been shown to cross-react with a number of different drug types most notably when using the EMIT II® kit (59, 60). Twelve patients with unexpected positive results for LSD were caused by the mucolytic drug ambroxol (58). The compound also exhibits a significant cross-reactivity in the CEDIA DAU LSD® assay (58). More recently, the CEDIA LSD UDS® was shown to be subject to false positives from sertraline (62). The authors spiked DFU with sertraline (ranging from 0 to 2 μg/mL) (Table VI). The results obtained from the urine samples fortified with sertraline showed that the CEDIA LSD® screening was positive when concentrations reached 1.5 μg/mL (62).
Conclusion
This literature review summarizes many reports that demonstrate false-positive immunoassay results can be caused by a variety of compounds. Common structural motifs account for some of these occurrences, but false-positive results can also be caused by dissimilar structures. It is important to remember that antibodies are binding to a three-dimensional structure. While some similarities can be seen between two-dimensional chemical structures of cross-reacting compounds, the absence of a similar chemical appearance does not rule out immunoassay cross-reactivity. It is also important to recognize that drug metabolites can also cause false-positive results. Simply adding the parent drug to DFU and demonstrating the absence of a positive result does not rule out ingestion of a particular drug as the cause of a false-positive result. Immunoassays provide useful clinical information, but should be viewed as ‘presumptive positive’ results until confirmed by an independent chemical technique such as GC–MS or liquid chromatography–tandem mass spectrometry.
Acknowledgments
The authors acknowledge the generous funding provided by Waters Corporation for the University of California, San Diego (UCSD) clinical chemistry fellowship program. The authors graciously acknowledge Dr Judith A. Stone (UCSD) for her input in preparation of this manuscript.